Validate process requirements by creating test templates then, run validation test, get results and repeat test as necessary.
Take users requirements, create test templates, execute test and review results. Results are cross referenced to actual test line and requirement.
Use in Waterfall, Agile or any project environment. Software developers can now deliver better code to clients.
Optimize your daily testing requirements to Standard and Ad Hoc processes, globally.


Embedded software in medical devices must be validated and verified to reduce public health risks

Biomedical engineers continue to develop a number of life-enhancing and life-saving technologies with the use of software components

Effective validation contributes significantly to assuring drug quality

Proper data collection is crucial to a successful quality system in clinical laboratories and clinical research

Federal regulations recommend that software validation and verification activities be conducted throughout the entire software life cycle

Remarkable progress in the field of medicine is leading to new discoveries that need to meet strict regulatory standards
"Our clients invest in safeguards that improve on their current quality systems. Our BTSValidation application helps clients validate any software or hardware implementation as an integral part of delivering a safe product or service to their customers." -Tim Busch, Chief Software Architect
Enter user requirements: This application allows for software and hardware validation. An engineer can enter requirements based on specifications and test create templates.
Create test template: Engineers no longer have to start from scratch when creating test templates. An engineer can create a test template to do line by line testing.
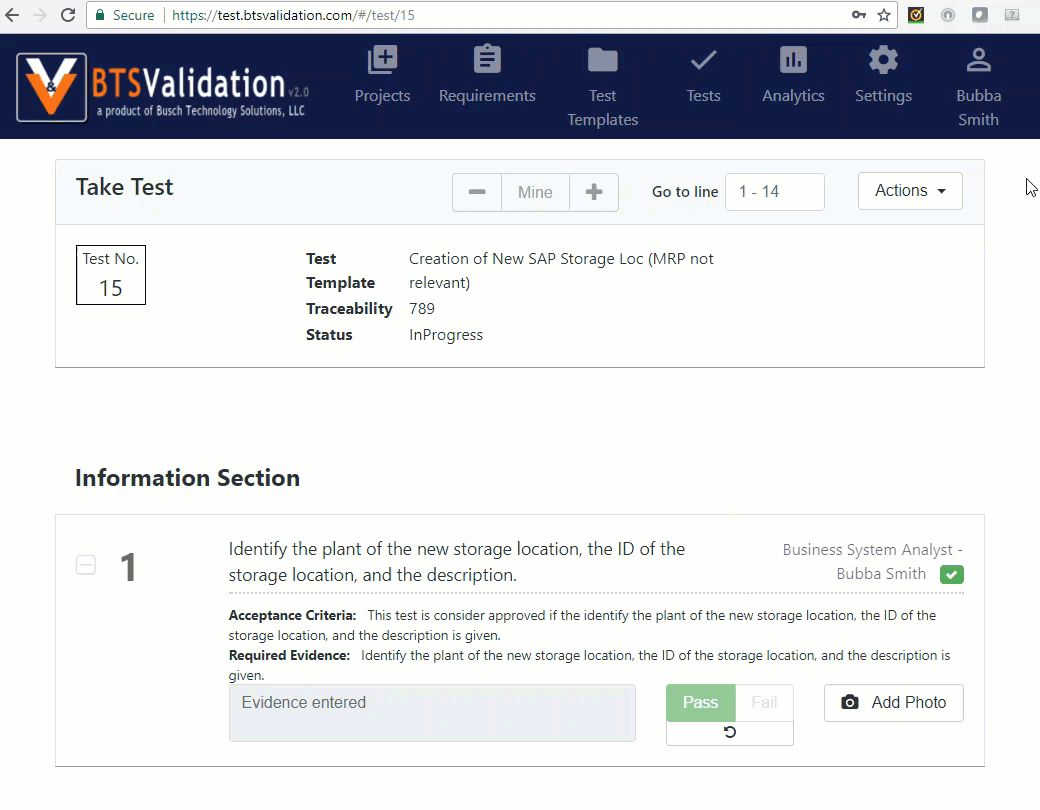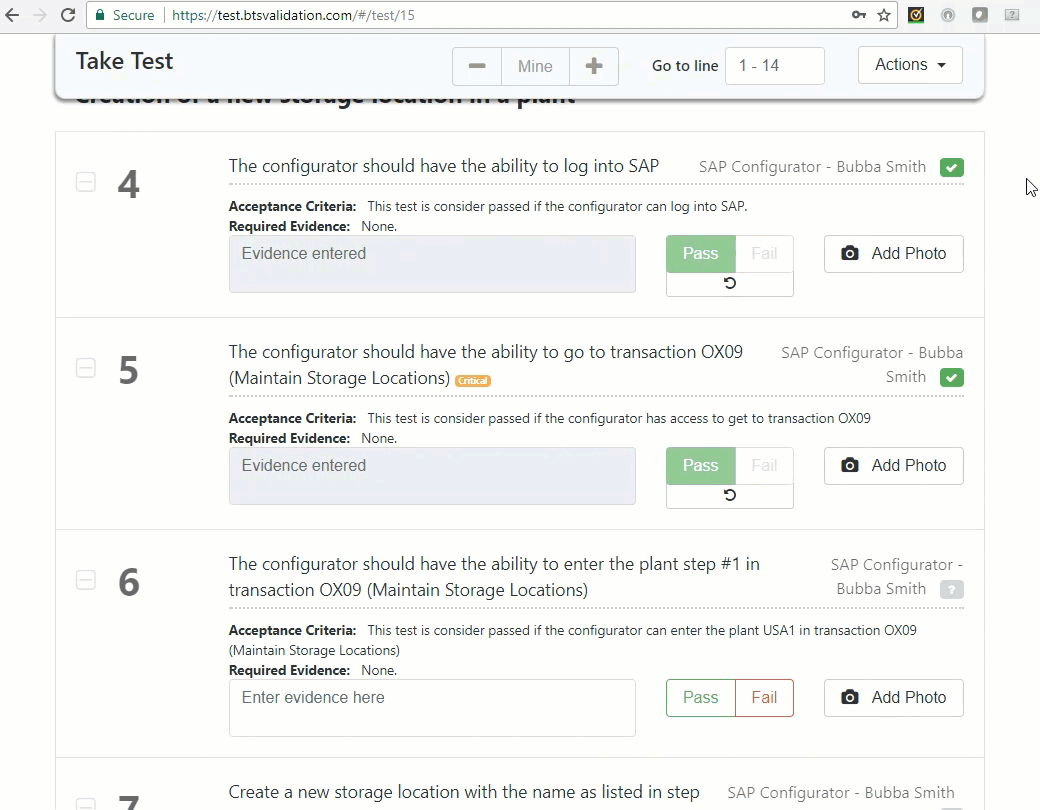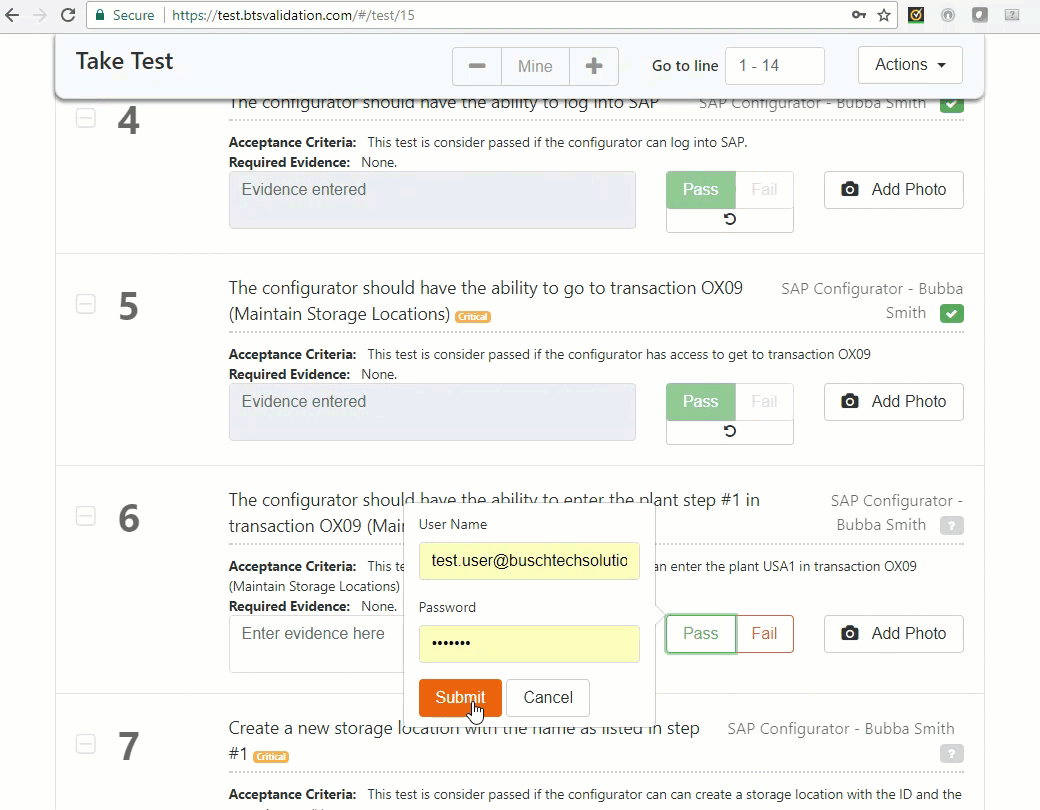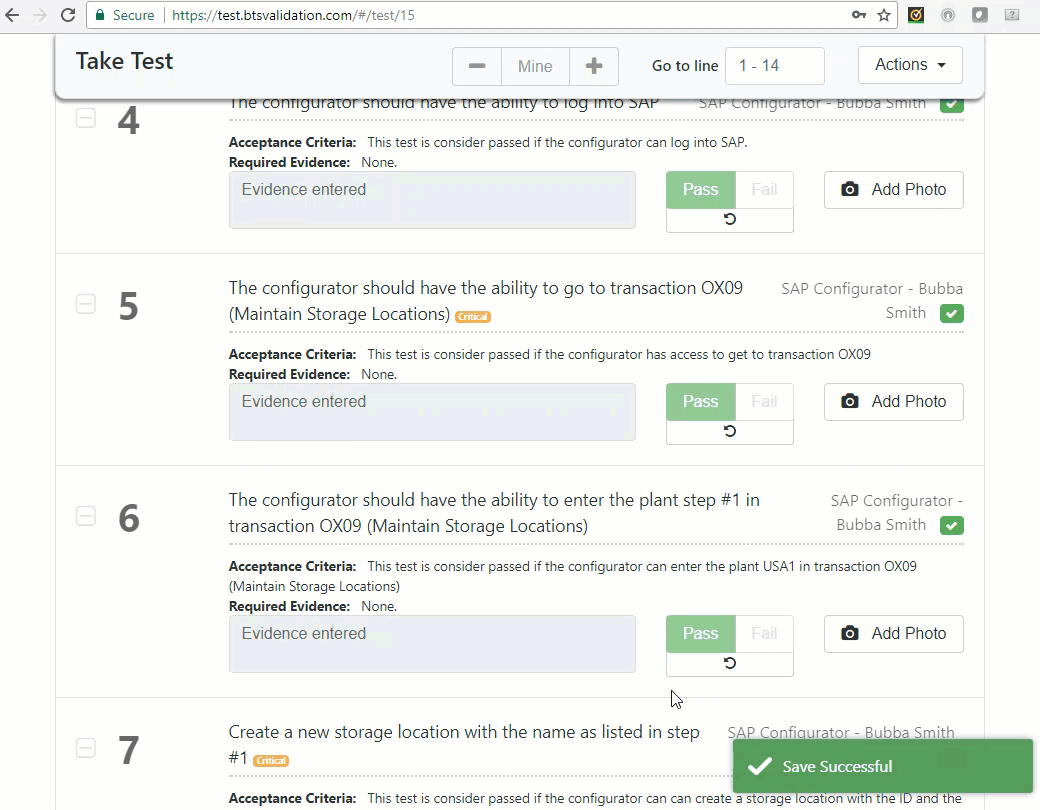
Execute test: An authorized user can take test and record findings.
Quality review: Critical lines and non-compliant lines can be addressed to make final decisions.
Generate report: The report contains cross references of test lines to their requirements and provides a Pass or Fail result. Print or save the test results.
The final report contains a cross reference of test line to its actual requirement and provides a Pass or Fail result per test line. This ensures traceability through entire test. It has never been this easy to show proof of proper validation & verification testing to management, finance or regulatory departments.

You have a 30-day free trial. Before the trial ends, we will give you the option of continuing your selected plan.
If you choose not to proceed, we will simply cancel your account login.